An in-depth look at thermal zinc diffusion



Thermal zinc diffusion, also known as sherardising or thermal diffusion galvanizing (TDG), is a well-established process to apply a protective zinc coating to steel parts. Unlike electroplating, which deposits a surface layer, thermal zinc diffusion creates a deeper, more permanent bond between the zinc and the steel through a diffusion process at elevated temperatures.
“This method offers superior corrosion resistance and durability, making it a valuable tool for various industries,” comments International Zinc Association (IZA) Africa Director Simon Norton. A number of South African firms offer zinc thermal diffusion, including Distek in Johannesburg.
Thermal zinc diffusion finds application in a range of industries due to its exceptional corrosion resistance properties. In the automotive industry, thermal zinc diffusion is used to protect fasteners, brackets, and other small automotive components from corrosion. In construction, threaded rods, screws, nails, and other hardware benefit from the long-lasting protection offered by thermal zinc diffusion.
In appliance manufacturing, components exposed to moisture, like washing machine components or refrigerator shelves, can be shielded from corrosion using thermal zinc diffusion. Threaded fittings, enclosures, and other electrical components can be protected from environmental factors using this process. Components used in harsh offshore environments like the oil and gas industry benefit from the superior corrosion resistance offered by thermal zinc diffusion.
The process commences with thorough cleaning of the steel parts. Any dirt, oil, or contaminants on the surface can hinder the diffusion process, so degreasing, grit blasting, or chemical cleaning is critical.
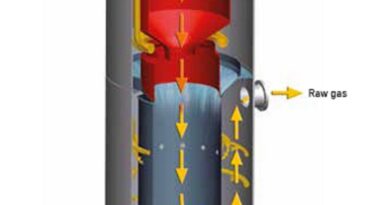
The cleaned parts are loaded into a tumbling barrel, or a static container filled with a specially formulated zinc powder. The powder plays a crucial role, as its composition can influence the final coating properties. Zinc powder variations include:
- Zinc dust: The most common type, a fine, grey powder produced by grinding zinc metal. Zinc dust offers a good balance between cost and performance.
- Zinc-aluminium mischmetal powder: A blend of zinc and aluminium. The addition of aluminium can enhance the corrosion resistance of the coating and improve its high-temperature performance.
- Pre-alloyed zinc powders: These powders contain specific elements pre-alloyed with zinc, offering tailored properties for specific applications. For example, adding rare earth elements can further enhance corrosion resistance.
The container is sealed and heated to a temperature range typically between 300°C to 430°C. The specific temperature selected depends on factors like the desired coating thickness and the type of zinc powder used. As the temperature increases, the zinc powder vapourises. These zinc vapours then come into contact with the hot steel surface.
The zinc atoms readily dissolve into the crystal structure of the steel to form a zinc-iron alloy layer on the surface. The depth of this layer depends on the process parameters like temperature and duration. The longer the parts are exposed to the high temperature, the deeper the diffusion, resulting in a thicker and more corrosion-resistant coating.
Once the required diffusion time is over, the container is cooled down. The cooling rate can also influence the final properties of the coating. The parts are removed, including any excess zinc powder. Depending on the application, a post-treatment like a passivation conversion coating or a paint layer might be applied for additional protection or aesthetics.
“Thermal zinc diffusion offers several advantages over other corrosion protection methods,” confirms Norton. The deep, diffused zinc layer offers excellent protection against rust and corrosion, even in harsh environments. This is because the zinc becomes an integral part of the steel, providing long-lasting protection.
The process also ensures a consistent and uniform coating, even on complex-shaped components with intricate details. The tumbling motion in barrels or the rotation of static containers allows for good contact between the zinc vapour and all surfaces of the parts.
As the zinc diffuses into the steel, there is minimal change in component dimensions, unlike electroplating, which can add a layer of material. It makes thermal zinc diffusion ideal for applications where precise tolerances are crucial.
Thermal zinc diffusion eliminates the need for hazardous chemicals like those used in electroplating. The zinc powder is typically the only material consumed in the process, making it a much eco-friendlier option.
The diffused zinc-iron layer is an integral part of the steel substrate, reducing the risk of flaking or peeling, even under harsh conditions. Thermal zinc diffusion coatings can last for decades, depending on the application and environmental conditions. The deep diffusion and excellent adhesion contribute to the long-lasting performance of these coatings.
“Thermal zinc diffusion is a highly effective method to protect steel components in a variety of industries,” highlights Norton. “Its ability to provide a deep, durable and environment friendly coating makes it an invaluable technology for ensuring the longevity and performance of critical steel parts,” he concludes.